Research Focus
(1) Hypoxia signaling mechanisms in Arabidopsis and Rice
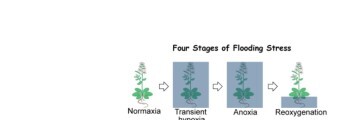
The long-term goal of this project is to elucidate the molecular mechanism by which plants respond to hypoxia stress caused by flooding in the soils. We found that cellular responses during flooding and post-flooding reoxygenation stages could determine the survival of plants under oxygen deprivation. Our studies showed that the phytohormone ethylene is a major signaling factor in regulating gene expression and metabolism. In addition, submergence triggers innate immunity in Arabidopsis via WRKY22, a response that may protect plants against pathogen infection either during or after flooding. Through phosphorproteomics, we found that the hypoxia signal triggers an SnRK1 phosphorylation cascade leading to the adjustment of anaerobic metabolism and selective translation of mRNAs of key hypoxic responsive genes (Fig. 1).
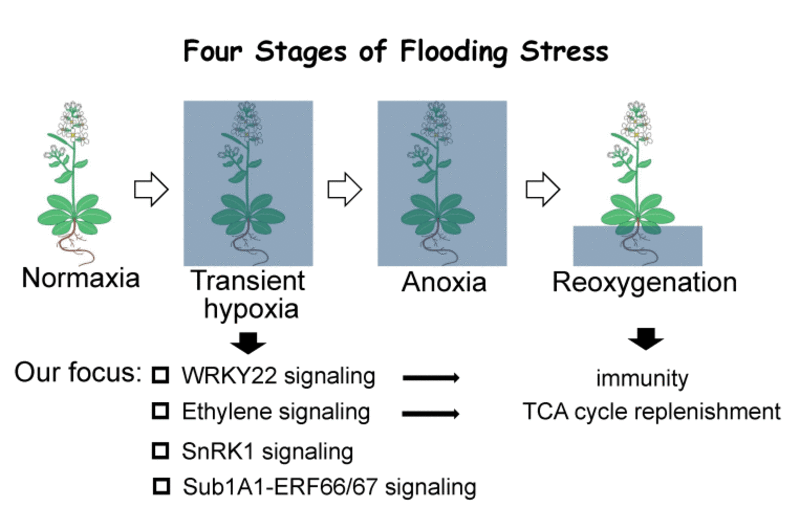
Based on these findings, our lab is currently conducting experiments to elucidate the molecular mechanism of SnRK1 in regulating translation dynamics in Arabidopsis under submergence. We are also investigating the functional roles of ethylene in regulating metabolic flow during hypoxia and reoxygenation (Fig. 2). In addition, we are interested in finding what is the signal that is triggered during early stages of hypoxia leading to the activation of downstream responsive genes such as WRKY22 and innate immunity genes. We have recently found that mutations in several members of lectin receptor-like kinases (LecRKs) gene family affect both hypoxia response and pathogen responses in Arabidopsis. We will conduct experiments to test whether the LecRK functions as the receptor to trigger immunity responses during hypoxia.
The rice Sub1A1 gene, which encodes a group VII ethylene response factor (ERFVII), is transcriptionally activated during submergence and plays a pivotal regulatory role in rice survival against flooding stress. One characteristic feature of ERFVIIs is a destabilizing N-terminus (Met-Cys-Gly-Gly) called the N-degron for the oxygen sensing mediated by the N-end rule pathway. Sub1A1 is the only known ERFVII with the conical N-degron sequence that can evade the oxygen-regulated N-end rule of protease degradation. We found that two other ERFVIIs, ERF66 and ERF67, are transcriptionally up-regulated by Sub1A1 under submergence. We also showed that ERF66 and ERF67 are substrates of N-end rule pathway of targeted proteolysis, so under hypoxia these two proteins might not just be the downstream of Sub1A-1 but also could be stabilized in a large amount to trigger hypoxia responses much stronger to allow rice survive from submergence. In summary, our results suggest that Sub1A1, ERF66 and ERF67 could form a regulatory cascade that involves transcriptional and N-end rule pathways in rice to tolerate submergence stress. We are currently investigating the molecular basis of how Sub1A1 can evade N-end rule regulation and elucidating the regulatory pathways mediated by the Sub1A1/ ERF66 and ERF67 cascade.
(2) Orchid functional genomics and biotechnology
The main goal of this project to construct genomic resources for Phalaenopsis aphrodite, which is an endemic species and a breeding parent for most white flower Phalaenopsis orchids in the markets. These resources will be used to build platforms for generation of new orchid breeds through molecular breeding and transgenic techniques.
By employing NGS (next generation sequencing) technology, our lab has generated a high quality draft of genomic sequences with linkage maps of P. aphrodite. We have combined NGS sequence assembly, BAC contig assembly and marker typing to generate a high quality draft genome with high coverage and low error rates (Fig. 3). Draft genomic sequences that conform to this standard are suitable general assessment of gene content and gene finding.

Currently, we are building transcriptomic and metabolomics resources for floral pigment and scent biosynthetic pathways as well as developmental pathways. We expect to employ these platforms to generate new breeds of Phalaenopsis orchids with shorter generation time, variable flower colors, scent and required shorter low temperature treatment.
(3) Exploring microbial genome for biotechnological applications
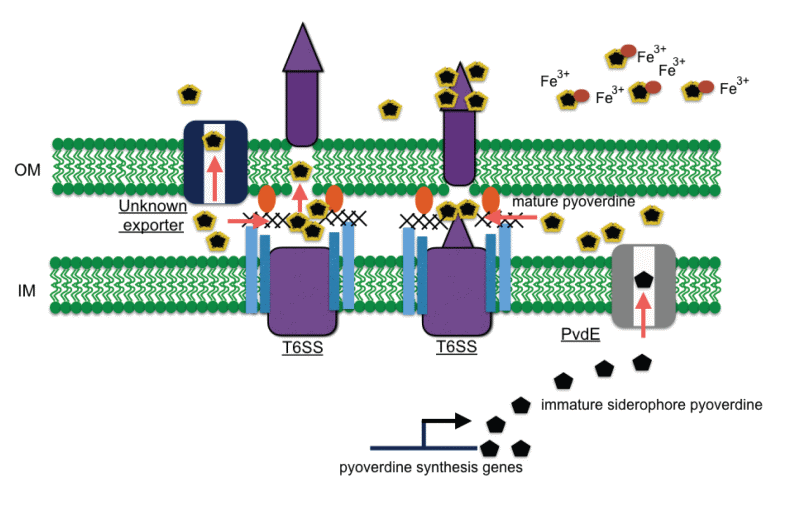
Pseudomonas taiwanensis is a novel Gram-negative bacterium, isolated from soil in Northern Taiwan using shrimp shell wastes as the sole carbon and nitrogen source. Using matrix-assisted laser desorption/ionization imaging mass spectrometry, we identified a novel pyoverdine secreted by P. taiwanensis that could inhibit growth of the rice blight pathogen Xanthomonas oryzae pv. oryzae (Xoo). Through Tn5 mutagenesis of P. taiwanensis, we showed that mutations in genes that encode components of type VI secretion system and biosynthesis and maturation of pyoverdine resulted in reduced toxicity against Xoo (Fig. 4).
We are developing P. taiwanesis and other microbes as sources for biocontrol agents. In addition to anti-rice bacterial blight disease, we have identified anti-fungal compounds and insecticidal proteins from P. taiwanese and other soil microbes. We will set up collaborations with biotech companies for field trial and to develop these compounds for farming applications.

Ming-Che Shih
Corresponding Research Fellow
- 2017 ~ Current, Distinguished Research Fellow and Chair, Academia Sinica Southern Campus Planning Commission
- 2016 ~ 2017, Distinguished Research Fellow and Secretary General of Academia Sinica
- 2008 ~ 2016, Distinguished Research Fellow and Director
- 2003 ~ 2008, Professor, Dept. of Biological Sciences, University of Iowa, USA
- 2005 ~ 2007, Director, Center for Comparative Genomics, University of Iowa, USA
- 1994 ~ 2003, Associate Professor, Dept. of Biological Sciences, University of Iowa, USA
- 1988 ~ 1994, Assistant Professor, Dept. of Biological Sciences, University of Iowa, USA
- 1984 ~ 1988, Postdoctoral Fellow Dept. of Genetics, Harvard Medical School, USA
- 1978 ~ 1983, Ph.D. Genetics Ph.D. Program, University of Iowa, USA